For Our Patients
Clinical Trials and Research
Enrolling in
a Study
Interested in Enrolling?
Thank you for your interest in participating in one of our clinical trials. These studies offer eligible new patients the opportunity to receive the latest, most advanced treatments for vitreoretinal conditions — all under the expert care of our dedicated clinical research team.
Currently Enrolling Studies
Last updated on Monday, March 25, 2026
If you’re a new patient and would like to be considered for enrollment, please click Enroll in this Study to send us an email. Be sure to include: Your full name, the reason you're interested in participating, the office location where you currently receive treatment. Our team will respond within 48–72 hours.
Below is a list of studies currently open for enrollment:
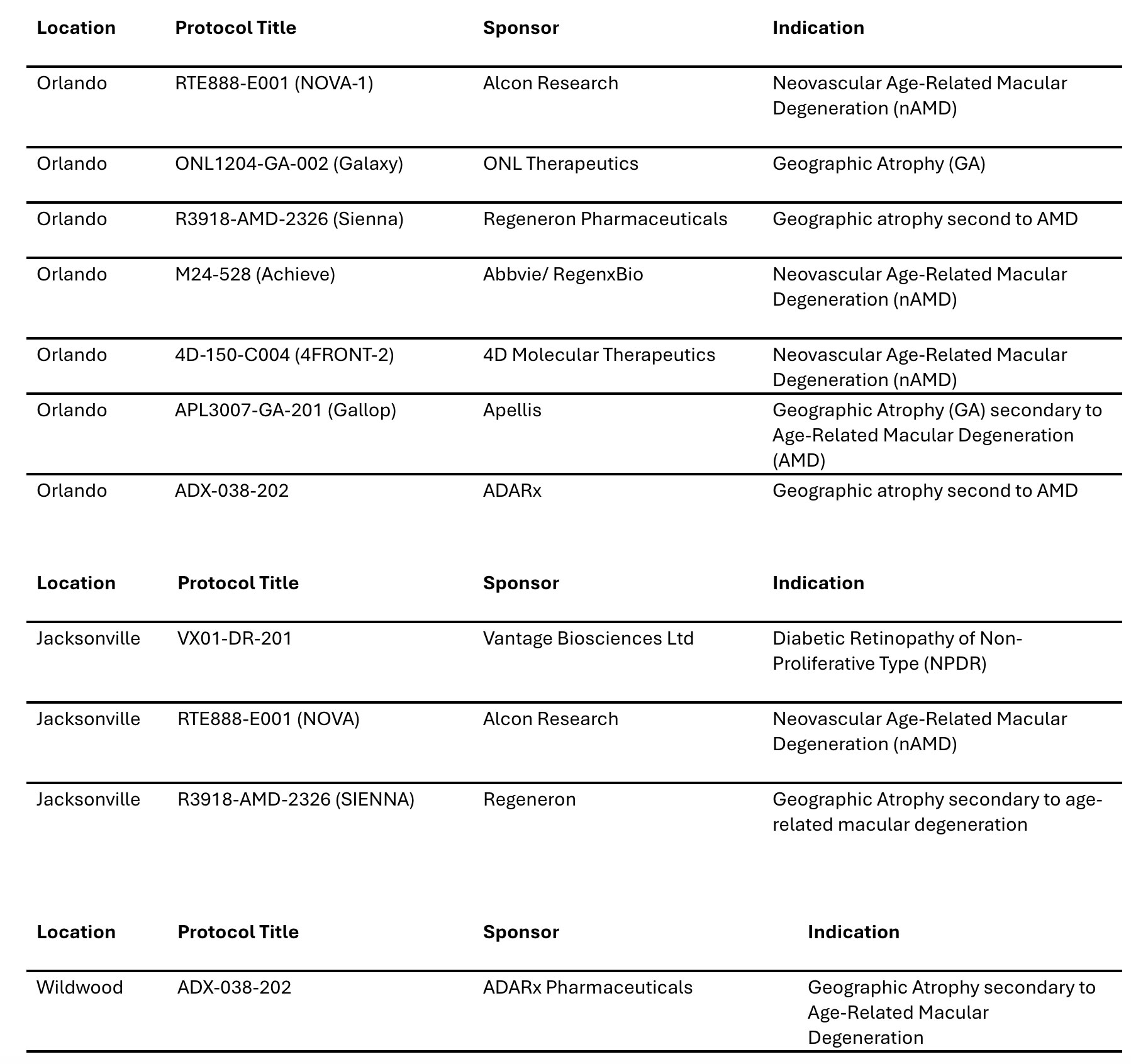
RESEARCH LOCATION: ORLANDO
Clinical Trial for Neovascular Age-Related Macular Degeneration (nAMD)
Protocol ID: RTE888-E001 (NOVA)
ClinicalTrials.gov ID: NCT05769153
Sponsor: Alcon Research
This Phase 1/2 two-stage dose-escalation study evaluates the safety, preliminary effectiveness, and durability of AR-14034 SR implant compared to intravitreal anti-VEGF (aflibercept), aiming to reduce injection frequency in patients with neovascular AMD.
Clinical Trial for Geographic Atrophy (GA)
Protocol ID: ONL1204-GA-002 (Galaxy)
ClinicalTrials.gov ID: NCT04744662
Sponsor: ONL Therapeutics
This trial investigates ONL1204, a Fas receptor antagonist, for its ability to slow photoreceptor cell death and disease progression in patients with Geographic Atrophy secondary to AMD.
Clinical Trial for Macular Neovascularization Secondary to AMD
Protocol ID: 4D-150-C003 (4Front-1)
ClinicalTrials.gov ID: NCT05481827
Sponsor: 4D Molecular Therapeutics
This Phase 2 trial evaluates 4D-150, an intravitreal gene therapy delivering aflibercept and anti-VEGF sequences, designed to provide long-term treatment for wet AMD and reduce injection burden.
Clinical Trial for Geographic Atrophy Secondary to AMD
Protocol ID: R3918-AMD-2326 (Sienna)
ClinicalTrials.gov ID: NCT06541704
Sponsor: Regeneron Pharmaceuticals
This Phase 3 randomized, double-masked, placebo-controlled trial examines the efficacy and safety of subcutaneously administered pozelimab—alone or with cemdisiran—to slow progression of Geographic Atrophy secondary to AMD.
Clinical Trial for Neovascular Age-Related Macular Degeneration (nAMD)
Protocol ID: M24-528 (Achieve)
ClinicalTrials.gov ID: NCT05890550
Sponsor: AbbVie / RegenxBio
This trial evaluates RGX-314, a gene therapy delivered via subretinal injection, aiming to provide sustained anti-VEGF expression and reduce treatment burden for nAMD patients.
Clinical Trial for Geographic Atrophy Secondary to AMD
Protocol ID: ADX‑038‑202
ClinicalTrials.gov ID: NCT06990269
Sponsor: ADARx Pharmaceuticals, Inc.
This Phase 2, randomized, masked, placebo‑controlled trial evaluates the efficacy, safety, pharmacokinetics (PK), and pharmacodynamics (PD) of subcutaneously administered ADX‑038 compared with placebo in participants with geographic atrophy (GA) secondary to age-related macular degeneration (AMD).
RESEARCH LOCATION: JACKSONVILLE
Clinical Trial for Macular Neovascularization Secondary to AMD
Protocol ID: 4D-150-C003 (4Front-1)
ClinicalTrials.gov ID: NCT05481827
Sponsor: 4D Molecular Therapeutics
This Phase 2 trial evaluates 4D-150, an intravitreal gene therapy delivering aflibercept and anti-VEGF sequences, designed to provide long-term treatment for wet AMD and reduce injection burden.
Clinical Trial for Diabetic Retinopathy of Non-Proliferative Type (NPDR)
Protocol ID: 1436-0007 (CRIMSON)
ClinicalTrials.gov ID: NCT05631270
Sponsor: Boehringer Ingelheim Pharmaceuticals Inc.
This trial investigates BI 1436, an oral therapy targeting pathways involved in diabetic retinopathy progression, aiming to prevent vision-threatening complications.
Clinical Trial for Diabetic Retinopathy of Non-Proliferative Type (NPDR)
Protocol ID: VX01-DR-201
ClinicalTrials.gov ID: NCT05890550
Sponsor: Vantage Biosciences Ltd
This study evaluates VX-01, a novel therapy intended to slow or reverse retinal vascular changes in NPDR patients.
Clinical Trial for Diabetic Macular Edema
Protocol ID: GR43828 (THAMES)
ClinicalTrials.gov ID: NCT06850922
Sponsor: Genentech, Inc.
This Phase I/II trial evaluates RO7446603—alone or co-administered with aflibercept or faricimab—to assess safety, tolerability, pharmacokinetics, and efficacy in participants with Diabetic Macular Edema.
Clinical Trial for Neovascular Age-Related Macular Degeneration (nAMD)
Protocol ID: RTE888-E001 (NOVA)
ClinicalTrials.gov ID: NCT05769153
Sponsor: Alcon Research
This Phase 1/2 two-stage dose-escalation study evaluates the safety, preliminary effectiveness, and durability of AR-14034 SR implant compared to intravitreal anti-VEGF (aflibercept), aiming to reduce injection frequency in patients with neovascular AMD.
Clinical Trial for Geographic Atrophy Secondary to AMD
Protocol ID: R3918-AMD-2326 (Sienna)
ClinicalTrials.gov ID: NCT06541704
Sponsor: Regeneron Pharmaceuticals
This Phase 3 randomized, double-masked, placebo-controlled trial examines the efficacy and safety of subcutaneously administered pozelimab—alone or with cemdisiran—to slow progression of Geographic Atrophy secondary to AMD.
RESEARCH LOCATION: WILDWOOD
Clinical Trial for Geographic Atrophy Secondary to Age-Related Macular Degeneration (GA secondary to AMD)
Protocol ID: ADX-038-202
ClinicalTrials.gov ID: NCT06990269
Sponsor: ADARx Pharmaceuticals
This Phase 2 randomized, masked, placebo-controlled study evaluates the efficacy, safety, pharmacokinetics, and pharmacodynamics of subcutaneously administered ADX-038, an investigational siRNA therapy, in patients with geographic atrophy secondary to age-related macular degeneration. The study is designed to assess whether systemic inhibition of complement factor B may slow the progression of GA lesions compared
to placebo, with the goal of addressing an unmet need in GA treatment
through a less invasive dosing approach than intravitreal therapies.
Studies closed for Enrollment
For more information about clinical trials or if you should have any questions, please feel free to contact us.




